
MyNIC®-S
Reducing Agent (Antioxidant)
Patented
Reducing agent (antioxidant) used by adding a small amount to tap or well water. It works by reducing the oxygen and chlorine content that can contribute towards oxidization to produce inexpensive, high-quality water with reducing power.
"Giving MyNIC®-S to Egg-laying Hens"
By adding just a small amount of MyNIC®-S to the drinking water of hens, they produce eggs with a more balanced nutritional content. This simple and easy method was proven to be highly effective.
→ Read the report here
MyNIC®-FM
Foliar Spray
Ministry of Agriculture, Forestry, and Fisheries registration
Liquid Compound Fertilizer
A foliar spray expected to have unprecedented and surprising effects in passing minerals through the cell membrane.
MyNIC®-C
Ion Cleaner
A unique cleaner combining cationic surfactants as well as Ca ions.
MyNIC®-A
Restoration Agent for Salt-Damaged Soil
Properties in Ca ions can be used to restore salt-damaged soil.

MyNIC-A|Properties in Ca ions can be used to restore salt-damaged soil.
Features of MyNIC®-A
The Ca ions that play a major part in the MyNIC® series have a "displacement" effect that can draw out harmful Na ions (sodium) from within salt-damaged soil and combines them with the chlorine ions present in the soil. This forms sodium chloride water, which penetrates deep into the soil where harmful salt does not affect the cultivation of crops.
Dilute 1.0 L of MyNIC®-A to 200 L by water and spray 15~17m2 of land to remove Na ions.
The stable Ca ion production method was developed by Olive Giken Ltd.
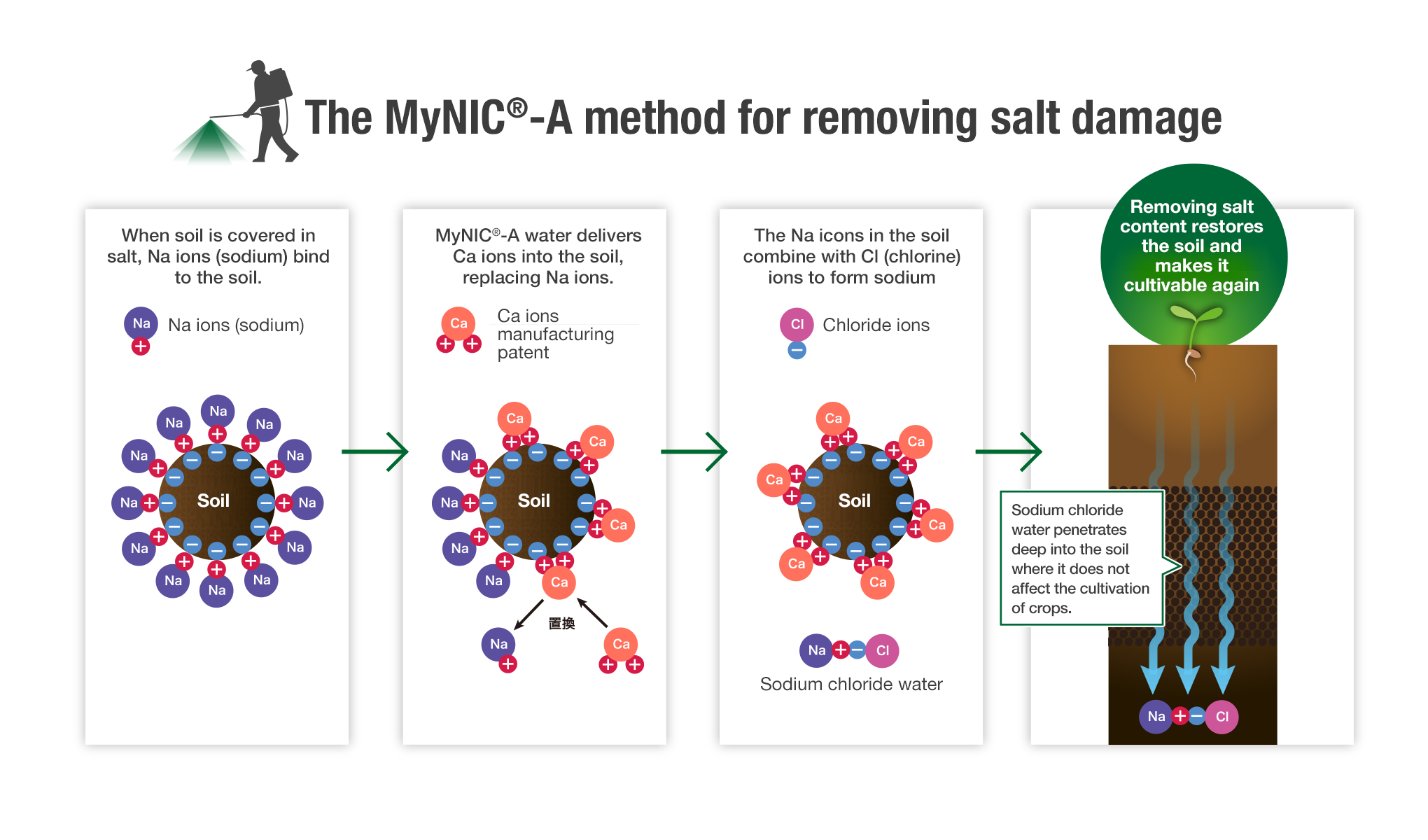
MyNIC®-A Overview
| Purpose |
|
||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| How to use | Dilute in 200 parts water and sprinkle on the affected soil, around 40L is used per tsubo (approx. 3.3m²) of soil. 1L of the solution should provide enough use for 50 tsubo (approx. 165m²) | ||||||||||||||||||||||||||||||||||||
| Mineral content | Ca ions | ||||||||||||||||||||||||||||||||||||
| Effects of Ca ions | Alkali metal ions such as Na ions bound to the soil colloid are replaced by Ca ions, returning the pH of the soil closer to a neutral level. | ||||||||||||||||||||||||||||||||||||
| Physical Properties |
The ORP and pH when the solution is diluted in tap water is as follows.
|
||||||||||||||||||||||||||||||||||||
| Stability | Stable for approximately 18 months as determined by ORP and O-NMR. It should be noted that while it did take on a brownish appearance, this is due to the oxidation of the plant extract added to stabilize Ca ions, and there is no effect on the actual Ca ions. | ||||||||||||||||||||||||||||||||||||

